Laboratory Focus

The focus of Dr. Mohan’s laboratory is to investigate epigenetic mechanisms, in particular, the role of microRNAs (miRNAs), long noncoding RNAs (lncRNAs) and DNA methylation in the molecular pathogenesis of HIV/SIV infection. Research in his laboratory is funded by three NIH grants, an R01 grant to study the role of epigenetic mechanisms regulating neuroinflammation in HIV infection. A second R01 focused on determining the role of extracellular vesicles in HIV induced gastrointestinal epithelial dysfunction and lymphoid fibrosis. A third R21/R33 grant will investigate the role of biomolecular condensates on HIV pathogenesis with a special focus on gastrointestinal and neurological dysfunction. Dr. Mohan has a broad background in veterinary medicine, pathology, and molecular biology, with specific training and expertise in key research areas pertaining to nonhuman primate immunology.
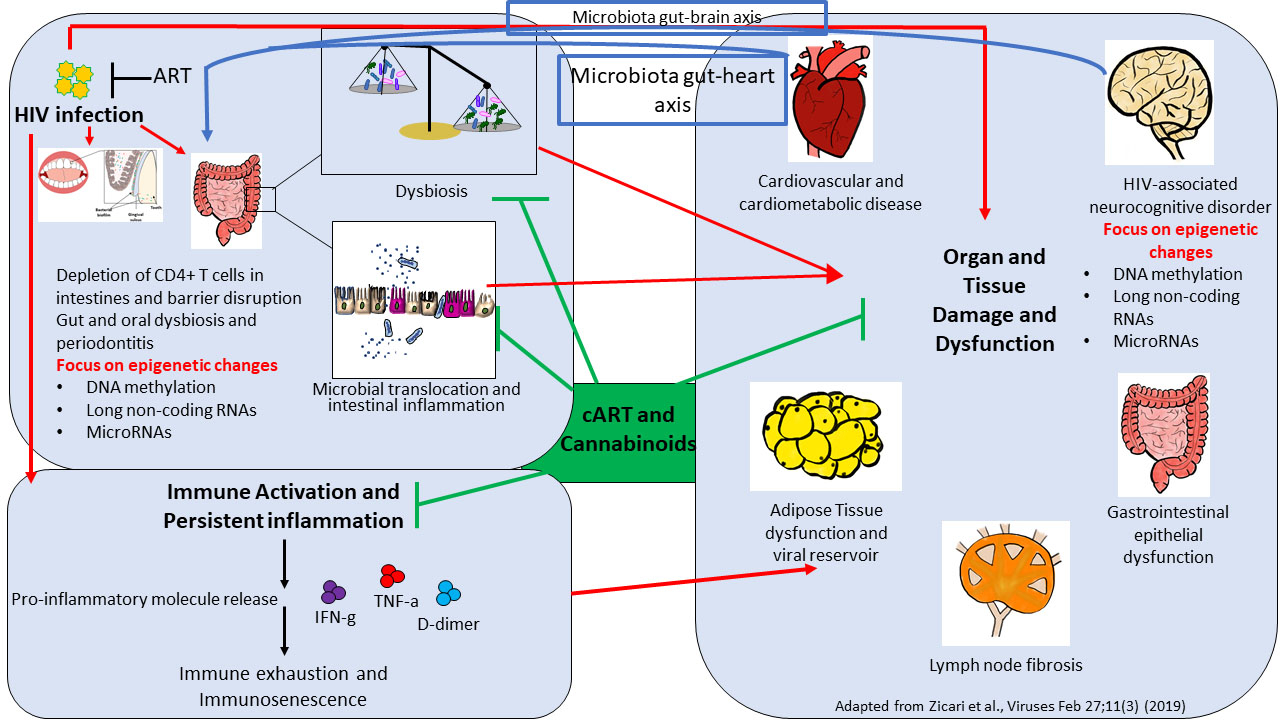
Although the advent of modern combination anti-retroviral therapy (ART) has increased the lifespans of people living with HIV (PLWH), they still have an elevated risk for developing non-AIDS related comorbidities such as cardiovascular disease, HIV associated neurocognitive disorders, metabolic diseases, etc. One of the factors driving these comorbidities is chronic inflammation/immune activation that persists in PLWH despite viral suppression by ART. To alleviate chronic inflammation, Dr. Mohan’s lab is currently investigating the combinatorial effect of suppressing viral replication and gastrointestinal/neuroinflammation using phytocannabinoids produced by the Cannabis sativa plant, namely, delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD), either alone or in combination on T and B-cell function, oral and intestinal microbiome (microbiota-gut-brain axis), plasma and cerebrospinal fluid metabolome and disease progression. His laboratory is extending the cannabinoid studies to other chronic inflammatory diseases, such as Tuberculosis and Alzheimer’s disease, using the unique rhesus macaque model of tuberculosis, and aged Baboon model of Alzheimer’s disease, respectively. Since 36 U.S. states (and more to follow) allow the use of cannabinoids for medical purposes, with citations of HIV/AIDS as a condition amenable to such treatment it is important to understand how THC and other phytocannabinoids regulate inflammation and disease progression in this population.
To study these inflammatory diseases and the effect cannabindoids can have on inflammation, we use the best biologically relevant models, rhesus macaques and baboons.
Laboratory Collaborations
Dr. Chioma Okeoma
Stonybrook University in New York
Dr. Siddappa Byrareddy
University of Nebraska, Omaha
Dr Jerzy Szablowski
Rice University
Dr. Jason Kimata
Baylor College of Medicine
Dr. Deepak Kaushal
Texas Biomed/SNPRC
Dr. Xavier Alvarez
Texas Biomed/SNPRC
Dr. Binhua Ling
Texas Biomed/SNPRC
Dr. Diako Ebrahimi
Texas Biomed
Dr. Smita Kulkarni
Texas Biomed

